- Blog
- Contre Jour Walkthrough
- Halogens
- Sly 2 Band Of Thieves All Cutscenes
- Vampire Diaries
- Pro Gamer Manager 2
- Barbearians Game
- Space Empires Iv Patch
- Youda Sushi Chef Same Dish Combo
- The Amazing Eternals Requirements
- Theatre Of Doom Walkthrough
- Euro Truck Simulator 2 Download Free Pc
- Auralux 2 Vergence Walkthrough

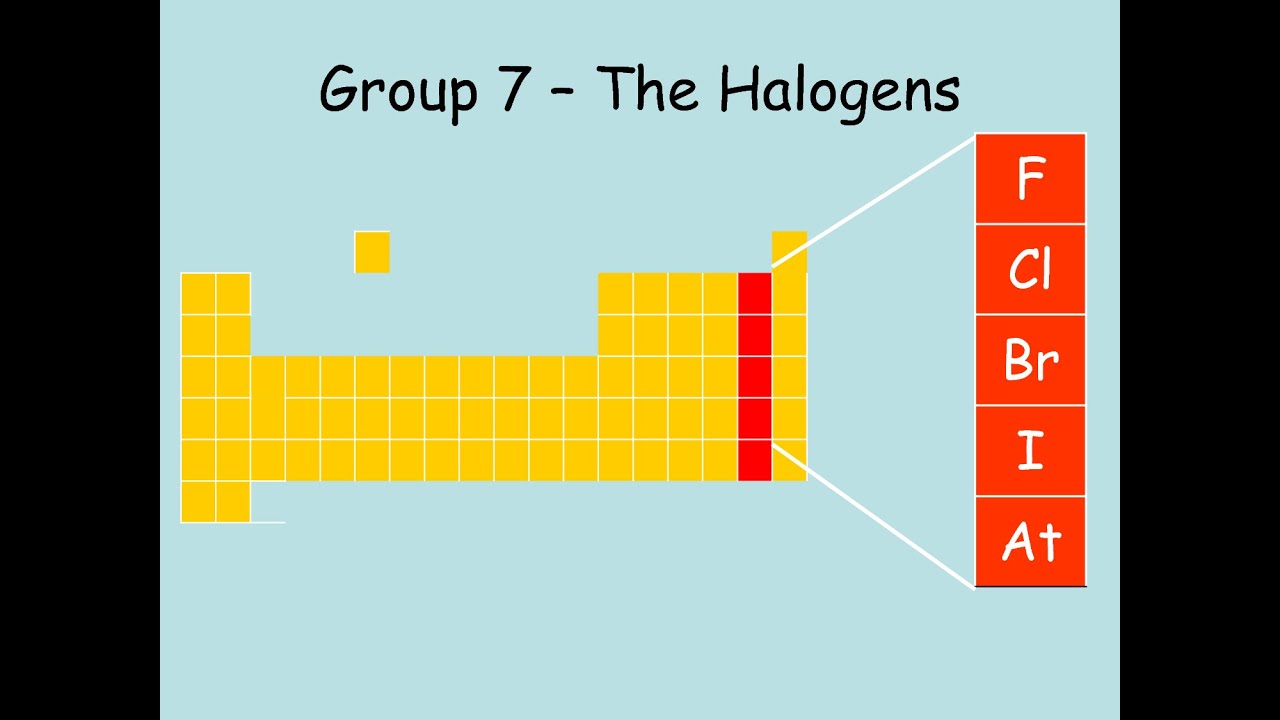
A halogen is a chemical element that forms a salt when it reacts with metal. Halogen lamps are illuminated by bulbs that contain a halogen and an inert gas.
Halogen, any of the six nonmetallic elements that constitute Group 17 (Group VIIa) of the periodic table. The halogen elements are fluorine (F), chlorine (Cl), bromine (Br), iodine (I), astatine (At), and tennessine (Ts). Learn more about the properties of halogens in this article. Halogens are a group of non-metallic elements that are placed in Group 17 of the Periodic Table. There are 5 elements that are considered to be a part of this group. These elements are Fluorine (F), Chlorine (Cl), Bromine (Br), Iodine (I), and Astatine (At).
There are five halogens in the periodic table of chemical elements: fluorine, chlorine, bromine, iodine, and astatine. The halogens are all highly reactive, which means they're quick to form bonds with other elements. What they often end up becoming is some kind of salt, including common table salt. In fact, the literal translation of halogen is 'salt-producer,' from Greek roots hals, 'salt,' and gen, 'giving birth to.'

